HanchorBio Presents Monotherapy and Combination Clinical Data for HCB101 at AACR Immuno-Oncology 2026
Monotherapy and multi-regimen combination data demonstrate a cytopenia-sparing safety profile, robust CD47 receptor occupancy, and encouraging dose-dependent antitumor activity
HanchorBio, Inc. (TPEx: 7827), a global clinical-stage biotechnology company pioneering transformative immunotherapies, today presented new clinical and translational data on HCB101, its differentiated SIRPα–Fc innate immune checkpoint fusion protein, at the American Association for Cancer Research (AACR) Immuno-Oncology Conference in Los Angeles. The presentation highlighted updated findings from the ongoing HCB101-101 Phase 1a monotherapy trial (NCT05892718) and the HCB101-201 Phase 1b/2a combination study (NCT06771622) across multiple tumor types and established oncology therapies.

The AACR Immuno-Oncology Conference is a focused international meeting dedicated exclusively to immuno-oncology science, convening leading academic investigators, translational researchers, and industry innovators to advance next-generation immune-based cancer therapies. Unlike broader oncology congresses, AACR-IO emphasizes mechanistic insights, translational strategies, and emerging clinical data across innate and adaptive immune pathways.
Presentation Details:
Application ID: 64087
Title: Phase 1 Monotherapy and Combination Data for HCB101, a Novel SIRPα-Fc Innate Checkpoint Fusion Protein
First Author: Dr. Fangling Ning, Affiliated Hospital of Binzhou Medical University
Date / Time: 19 February 2026 / 12:15 – 15:15 PT
Key Findings Highlighted
Phase 1a Monotherapy (HCB101-101; NCT05892718)
As of January 2026, 67 patients were enrolled across 13 dose levels (0.08 – 36 mg/kg QW):
- Clean, cytopenia-sparing safety across dose levels
- No bleeding events or immune-related toxicities; predominantly Grade 1-2 treatment-related adverse events
- 2 dose-limiting toxicities (thrombocytopenia), both resolved
- Maximum tolerated dose not reached
- Linear PK (t1/2 ~3 days) with receptor occupancy (RO) >99% at ≥8 mg/kg
- Antitumor activity, including confirmed partial responses in:
- Head and neck squamous cell carcinoma (~42% tumor regression, ≥32 weeks)
- Marginal zone lymphoma (~89% tumor regression, ≥16 weeks)
- Durable stable disease (≥4-9 months) across colorectal, ovarian, non-small cell lung cancer, and sarcoma
Combination Therapy (HCB101-201; NCT06771622)
HCB101 is being evaluated with chemotherapy, PD-1, anti-VEGF, HER2-targeted, and EGFR-based regimens. Across combination cohorts:
- Well-tolerated across gastric cancer (GC), triple-negative breast cancer (TNBC), colorectal cancer (CRC), and HNSCC
- No unexpected overlapping toxicities or new safety signals
- Cytopenias are primarily attributable to chemotherapy rather than HCB101
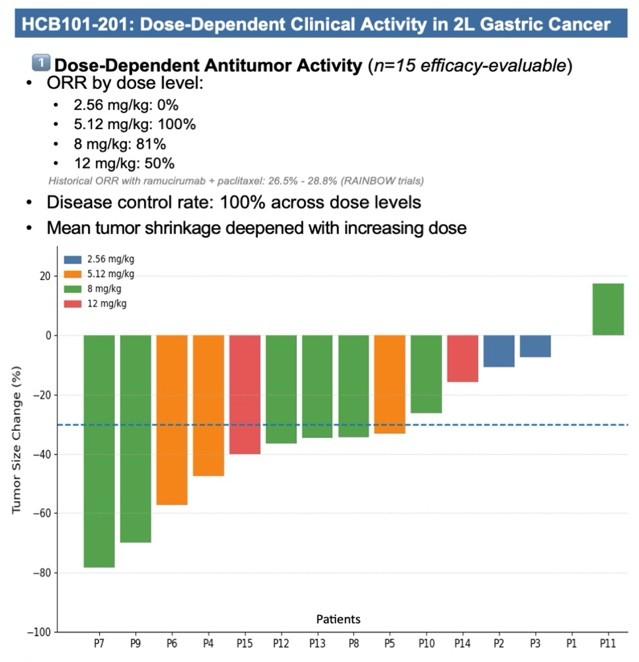
- 2L GC (Ramucirumab + Paclitaxel):
- ORR: 100% (5.12 mg/kg) and 81% (8 mg/kg)
- Disease control rate (DCR): 100% across dose levels
- Dose-dependent tumor shrinkage, with deepest reduction up to -78.2%
- 1L GC (HER2+ backbone): ORR up to 100% in early cohorts; DCR 100%
- 1L TNBC: ORR up to 67% in early cohorts; DCR 100%
“Across both monotherapy and combination settings, HCB101 is demonstrating a consistent and differentiated clinical profile – cytopenia-sparing safety, sustained receptor occupancy, and early dose-dependent activity signals,” said Alvin Luk, PhD, MBA, CCRA, President & Chief Medical Officer (Group) and CEO (U.S.A.) of HanchorBio. “The observed activity, particularly in combination regimens, strengthens our confidence in positioning HCB101 as a macrophage-checkpoint backbone. Our objective is not incremental refinement of CD47 biology but to establish a reliable innate immune foundation that integrates seamlessly with established oncology standards of care. The data presented at AACR-IO reinforce that direction and provide a strong basis for ongoing Phase 2 development in tumor types with high unmet need.”
About HCB101: A Next-Generation SIRPα Fc-Fusion Protein
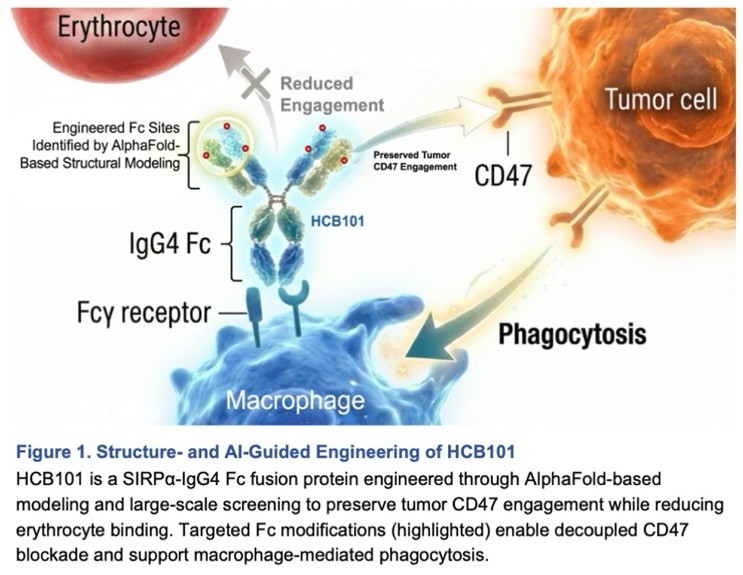
HCB101 is a rationally engineered SIRPα–IgG4 Fc fusion protein developed using HanchorBio’s FBDB™ platform to selectively block the CD47–SIRPα innate immune checkpoint while minimizing hematologic toxicity. Unlike earlier anti-CD47 approaches, HCB101 is designed to preserve macrophage-mediated antitumor activity while reducing red blood cell binding, a limitation that historically constrained CD47-directed therapies.
Engineered using AI-assisted structural modeling, HCB101 achieves differentiated binding to CD47 on tumor cells while maintaining low affinity for CD47 on red blood cells. Its safety, receptor occupancy, and pharmacologic characteristics are designed to enable integration with established oncology regimens without disrupting standard dosing or clinical workflows.
In ongoing clinical and translational evaluation, HCB101 has demonstrated sustained target engagement and early antitumor activity as both monotherapy and in combination, including in tumor types historically challenging for CD47-directed therapies. Dose escalation and Phase 2 expansion cohorts in gastric, colorectal, and head and neck cancers are ongoing.
These attributes support HCB101’s development as a differentiated innate immune checkpoint backbone with broad potential across solid tumors and hematologic malignancies.
About HanchorBio
Based in Taipei, Shanghai, and the San Francisco Bay Area, HanchorBio (7827.TPEx) is a global clinical-stage biotechnology company focused on immuno-oncology and immune-mediated diseases. The company is led by an experienced team with a proven track record in biologics discovery and global development, with the aim of reshaping the landscape of cancer therapies. HanchorBio’s proprietary Fc-based designer biologics (FBDB™) platform enables the design of multi-functional biologics with diverse targeting modalities, designed to activate both innate and adaptive immune pathways and overcome the current challenges of anti-PD1/L1 immunotherapies. The FBDB™ platform has delivered proof-of-concept data in several in vivo tumor animal models. HanchorBio is advancing a portfolio of innovative biologics designed to address significant unmet medical needs through differentiated molecular configurations in R&D and scalable CMC strategies.
漢康生技於美國癌症研究協會 (AACR) 腫瘤免疫學年會發表 HCB101 單藥與聯合治療的臨床數據
單藥與多重聯合治療數據顯示具「避免血球細胞減少」之安全性、穩定且高度的 CD47 受體佔據率 (RO),以及具劑量依賴性的抗腫瘤活性
專注於開發創新癌症與自體免疫疾病免疫療法的全球臨床階段生物技術公司——漢康生技(HanchorBio, Inc.;證券代碼:7827),19日於美國癌症研究協會 (AACR)腫瘤免疫學年會(AACR Immuno-Oncology Conference)中,發表其差異化先天免疫檢查點融合蛋白 HCB101 的臨床進展與轉譯醫學數據。
本次發表重點包含正在進行中的 HCB101-101 第一階段單藥臨床試驗 (NCT05892718) 以及 HCB101-201 第1b/2a期聯合治療臨床試驗 (NCT06771622) 之更新結果,涵蓋多種腫瘤類型與既有標準癌症療法聯合應用之成果。
AACR 免疫腫瘤學年會為專注於免疫腫瘤學科學的國際會議,匯集頂尖學術與產業專家,聚焦於先天與後天免疫機制及其臨床轉譯突破。
與綜合性腫瘤年會不同,AACR免疫腫瘤學年會特別著重於機制探討、轉譯策略及先天與後天免疫路徑之臨床新興數據。
發表資訊
- Application ID: 64087
- 論文標題: Phase 1 Monotherapy and Combination Data for HCB101, a Novel SIRPα-Fc Innate Checkpoint Fusion Protein
- 第一作者: 寧方玲醫師(濱州醫學院附屬醫院)
- 發表時間: 2026 年 2 月 19 日 12:15–15:15(太平洋時間)
重點研究成果
一、第一期單藥試驗(HCB101-101;NCT05892718)
截至 2026 年 1 月,共納入 67 位受試者,涵蓋 13 個劑量梯度(0.08–36 mg/kg,每週一次):
- 各劑量組皆展現清晰且避免細胞減少(cytopenia-sparing)的安全特徵
- 未觀察到出血事件或免疫相關毒性;治療相關不良事件多為第 1–2 級
- 發生 2 例劑量限制性毒性(血小板減少),皆已恢復
- 尚未達到最大耐受劑量(MTD)
- 呈現線性藥物動力學(半衰期約 3 天),於 ≥8 mg/kg 劑量下受體佔據率(RO)>99%
- 觀察到抗腫瘤活性,包括以下已確認之部分緩解(PR):
- 頭頸部鱗狀細胞癌(腫瘤縮小約 42%,持續 ≥32 週)
- 邊緣區淋巴瘤(腫瘤縮小約 89%,持續 ≥16 週)
- 多種腫瘤類型觀察到持續性疾病穩定(4–9 個月以上),包括:
- 大腸直腸癌
- 卵巢癌
- 非小細胞肺癌
- 肉瘤
二、聯合治療試驗(HCB101-201;NCT06771622)
HCB101 正與化療、PD-1 抑制劑、抗 VEGF、HER2 標靶療法及 EGFR 為基礎之療法進行合併評估。
整體合併治療結果顯示:
- 於胃癌(GC)、三陰性乳癌(TNBC)、大腸直腸癌(CRC)與頭頸癌(HNSCC)族群中皆具良好耐受性
- 未觀察到預期外的重疊毒性或新的安全性訊號
- 血球減少主要歸因於化療,而非 HCB101
二線胃癌(Ramucirumab + Paclitaxel)
- 客觀緩解率(ORR):
- 5.12 mg/kg:100%
- 8 mg/kg:81%
- 疾病控制率(DCR):各劑量組皆為 100%
- 顯示劑量依賴性腫瘤縮小,最大縮小幅度達 -78.2%
一線胃癌(HER2 陽性標準療法合併)
- 初期族群 ORR 最高達 100%
- DCR 為 100%
一線三陰性乳癌(TNBC)
- 初期族群 ORR 最高達 67%
- DCR 為 100%
漢康生技總裁暨醫療長、暨美國子公司執行長 陸英明博士表示:
「無論在單藥或聯合治療情境下,HCB101 均展現出一致且具差異化的臨床特性—不易引發血球減少的安全性、持續性的高受體佔據率,以及早期劑量依賴性活性訊號。
特別是在合併療法中的療效表現,更強化我們將 HCB101 定位為『巨噬細胞檢查點骨幹療法』的信心。我們的目標並非僅是改良 CD47 生物學,而是建立一個可與現行標準治療無縫整合的可靠先天免疫基礎。
AACR-IO 所發表的數據再次印證此一發展方向,並為後續在高度未被滿足醫療需求之腫瘤類型中推進第二期臨床試驗奠定堅實基礎。」
關於 HCB101: 新一代 SIRPα Fc 融合蛋白
HCB101 是一款經由理性設計的 SIRPα–IgG4 Fc 融合蛋白,基於漢康生技專有的 FBDB™ 平台開發,旨在選擇性阻斷 CD47–SIRPα 先天免疫檢查點,同時將血液學毒性降至最低。不同於早期的抗 CD47 研發路徑,HCB101 的設計旨在保留巨噬細胞介導的抗腫瘤活性,並減少對紅血球的結合;對紅血球的結合過去一直是限制 CD47 標靶療法臨床應用的一大瓶頸。
HCB101 利用 AI 輔助結構建模技術,實現了對癌細胞上 CD47 的差異化結合,同時對紅血球上的 CD47 保持低親和力。其安全性指標、受體佔位特性及藥理屬性,旨在支持其與現有腫瘤治療方案的整合,且不影響標準給藥劑量、安全預期或臨床作業流程。在目前的臨床與轉化研究評估中,HCB101 作為單藥及聯合治療均展現出穩定的標靶結合能力及早期抗腫瘤活性,其涵蓋的腫瘤類型甚至包括過去被認為對 CD47 標靶療法極具挑戰性的癌種。
綜合上述特質,HCB101 已定位為具備差異化的先天免疫檢查點骨幹(Backbone),在實體瘤與血液惡性腫瘤的各類聯合治療策略中展現出廣闊的應用潛力。
關於漢康生技(HanchorBio)
漢康生技(證券代碼:7827.TPEx)總部位於台北、上海及舊金山灣區,是一家專注於免疫腫瘤學及免疫媒介疾病的全球臨床階段生物技術公司。公司由擁有豐富藥物研發與全球臨床開發實戰經驗的資深團隊領軍,致力於重塑癌症治療版圖。
漢康生技致力於重啟人體免疫系統以對抗疾病,其專有的 Fc 基礎設計生物藥(FBDB™)平台能設計出具備多種標靶模式的獨特多功能生物藥,旨在激發先天與後天免疫通路,以克服現有 anti-PD-1/L1 免疫療法的瓶頸。FBDB™ 平台已在多個體內(in vivo)腫瘤動物模型中成功取得概念驗證數據。透過在多功能生物藥研發領域的突破,以及具備高擴展性的 CMC(化學製造與管制)策略,漢康生技正加速推進創新藥物管線,以解決尚未被滿足的重大醫療需求。



