HanchorBio Presents Dose-Dependent Tumor Responses and Deepening Tumor Shrinkage with HCB101 Triple Combination in Second-Line Gastric Cancer at the 11th Asia-Pacific Gastroesophageal Cancer Congress
Early clinical data show 100% ORR at 5.12 mg/kg, 100% DCR across doses, and dose-dependent tumor shrinkage; the 12 mg/kg cohort remains ongoing

HanchorBio, Inc. (TPEx: 7827), a global clinical-stage biotechnology company advancing transformative immunotherapies, today presented new clinical data from its ongoing HCB101-201 Phase 1b/2a study (NCT06771622) at the 11th Asia-Pacific Gastroesophageal Cancer Congress (APGCC) in Okinawa, Japan. The poster highlights early findings supporting dose-dependent antitumor activity of HCB101 in combination with ramucirumab and paclitaxel in patients with advanced gastric adenocarcinoma who progressed after first-line therapy.
The dataset demonstrates dose-dependent tumor reduction and consistent disease control across different dose levels, supporting further evaluation of HCB101 as a differentiated innate immune checkpoint backbone that integrates with standard-of-care regimens.
As previously announced in February 2026, HCB101 has been granted Orphan Drug Designation by the U.S. Food and Drug Administration (FDA) for the treatment of gastric cancer.
Poster Presentation Details
Title: Phase Ib/IIa Study of HCB101 Combination Demonstrates Striking Dose-Dependent Efficacy in Second-Line Gastric Cancer: 100% Partial Responses at 5.12 mg/kg
Presenter: Abigail Yu
Key Findings Highlighted
The poster reports dose-dependent antitumor activity of HCB101 when combined with ramucirumab and paclitaxel in second-line gastric cancer, including both response rates and tumor shrinkage depth.
Efficacy (15 efficacy-evaluable patients across dose levels)
- Objective response rate (ORR) by dose:
- 56 mg/kg: 0%
- 12 mg/kg: 100%
- 8 mg/kg: 81%
- 12 mg/kg (ongoing): preliminary ORR observed to date
- Disease control rate (DCR): 100% across all doses
- Mean tumor shrinkage by dose:
- 56 mg/kg: –6.0%
- 12 mg/kg: –33.4%
- 8 mg/kg: –46.0%
- 12 mg/kg: –27.9% (data continue to mature)
Overall, the dataset supports a dose-dependent efficacy profile, with tumor reductions deepening through the mid-to-high dose range; interpretation of the 12 mg/kg cohort remains preliminary due to ongoing enrollment and follow-up.
Safety
- No dose-limiting toxicities were observed in DLT-evaluable patients
- The most frequently reported treatment-related adverse events were:
- decreased in white blood cell count (leukopenia)
- decreased in neutrophil count (neutropenia)
Overall, hematologic adverse events were manageable with standard supportive care. No unexpected safety signals were observed in the current study. The most common events (leukopenia and neutropenia) were consistent with the known effects of chemotherapy, and the overall safety profile was consistent with expectations.
“HCB101 is engineered to selectively target the CD47–SIRPα innate immune checkpoint while remaining compatible with established standard oncology regimens,” said Alvin Luk, PhD, MBA, CCRA, President & Chief Medical Officer (Group) and CEO (USA) of HanchorBio. “At APGCC, we reported clear dose-dependent activity in second-line gastric cancer, including confirmed responses at 5.12 mg/kg and deeper tumor shrinkage at 8 mg/kg. As higher-dose cohorts continue to mature, these data reinforce our strategy to develop HCB101 as a differentiated innate immune backbone that integrates seamlessly with standard-of-care therapies.”
Combination Rationale
The regimen is designed to coordinate complementary mechanisms within the tumor microenvironment: HCB101 restores macrophage-mediated phagocytosis by selectively blocking the CD47–SIRPα innate immune checkpoint; ramucirumab mitigates VEGF-driven immunosuppression and supports vascular normalization; and paclitaxel promotes immunogenic tumor cell death and antigen release. Together, the combination aims to enhance both innate and adaptive antitumor responses within a standard-of-care backbone.
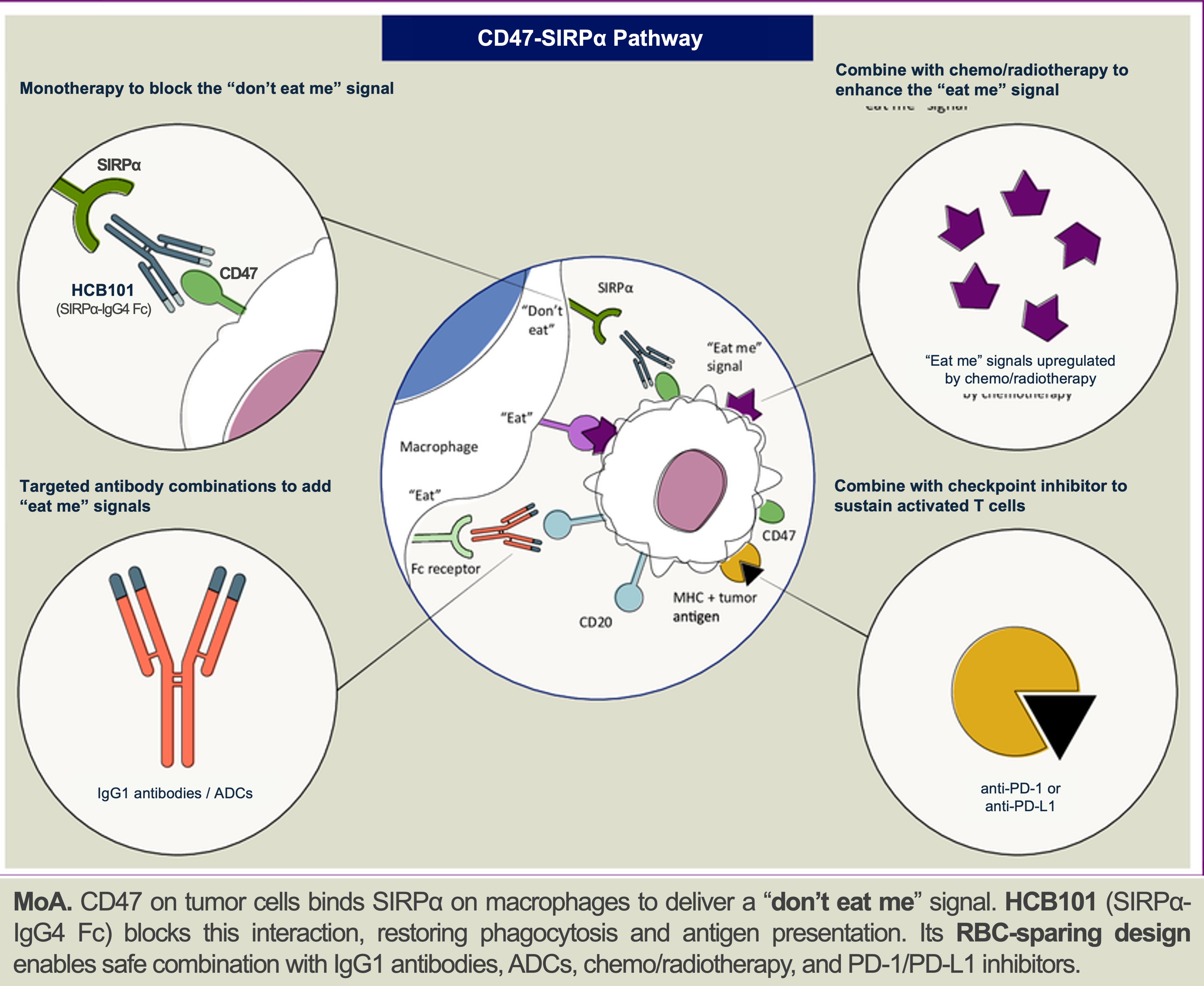
About HCB101: A Next-Generation SIRPα Fc-Fusion Protein
HCB101 is a rationally engineered SIRPα–IgG4 Fc fusion protein developed using HanchorBio’s FBDB™ platform to selectively block the CD47–SIRPα innate immune checkpoint while minimizing hematologic toxicity. Unlike earlier anti-CD47 approaches, HCB101 is designed to preserve macrophage-mediated antitumor activity while reducing red blood cell binding, a limitation that historically constrained CD47-directed therapies.
Engineered using AI-assisted structural modeling, HCB101 achieves differentiated binding to CD47 on tumor cells while maintaining low affinity for CD47 on red blood cells. Its safety, receptor occupancy, and pharmacologic characteristics are designed to enable integration with established oncology regimens without disrupting standard dosing or clinical workflows.
In ongoing clinical and translational evaluation, HCB101 has demonstrated sustained target engagement and early antitumor activity as both monotherapy and in combination, including in tumor types historically challenging for CD47-directed therapies. Dose escalation and Phase 2 expansion cohorts in gastric, colorectal, head-and-neck, and triple-negative breast cancers are ongoing.
These attributes support HCB101’s development as a differentiated innate immune checkpoint backbone with broad potential across solid tumors and hematologic malignancies.
About HanchorBio
Based in Taipei, Shanghai, and the San Francisco Bay Area, HanchorBio (7827.TPEx) is a global clinical-stage biotechnology company focused on immuno-oncology and immune-mediated diseases. The company is led by an experienced team with a proven track record in biologics discovery and global development, aiming to reshape the landscape of cancer therapies. HanchorBio’s proprietary Fc-based designer biologics (FBDB™) platform enables the design of multi-functional biologics with diverse targeting modalities, designed to activate both innate and adaptive immune pathways and overcome the current challenges of anti-PD1/L1 immunotherapies. The FBDB™ platform has delivered proof-of-concept data in several in vivo tumor animal models. HanchorBio is advancing a portfolio of innovative biologics designed to address significant unmet medical needs through differentiated molecular configurations in R&D and scalable CMC strategies.
漢康生技於第11屆亞太胃食管癌大會(APGCC)發表 HCB101 三藥聯合療法於二線胃癌之具劑量依賴性的腫瘤反應與持續腫瘤縮小數據
早期臨床數據顯示:5.12 mg/kg 劑量達到 100% 客觀緩解率(ORR),各劑量組疾病控制率(DCR)達 100%,且腫瘤縮小呈劑量依賴趨勢;12 mg/kg 劑量組仍持續收案中
漢康生技為致力於開發突破性免疫療法的全球臨床階段生技公司。公司今日於日本沖繩舉行的第11屆亞太胃食管癌大會(Asia-Pacific Gastroesophageal Cancer Congress, APGCC)發表其正在進行中的 HCB101 臨床 1b/2a期臨床試驗(NCT06771622)最新臨床數據。
壁報展示了公司自主開發的HCB101 與及 paclitaxel 聯合治療,用於一線治療後疾病進展之晚期胃腺癌患者中的早期研究結果,顯示出劑量依賴性的抗腫瘤活性。
本次資料顯示,HCB101在不同劑量組中呈現劑量依賴性的腫瘤縮小,且各劑量組皆觀察到一致的疾病控制,支持持續評估HCB101 作為一種具差異化的先天免疫檢查點骨幹(innate immune checkpoint backbone)療法,並可與現行標準治療方案整合。
此外,公司已於 2026 年 2 月公告,HCB101 獲 美國食品藥物管理局(FDA)授予用於治療胃癌的孤兒藥資格認定(Orphan Drug Designation)。
海報發表資訊
題目
Phase Ib/IIa Study of HCB101 Combination Demonstrates Striking Dose-Dependent Efficacy in Second-Line Gastric Cancer: 100% Partial Responses at 5.12 mg/kg
發表者
Abigail Yu
主要研究結果
本研究報告 HCB101 與 ramucirumab 及 paclitaxel 聯合療法在二線胃癌治療中呈現劑量依賴性的抗腫瘤活性,包括緩解率與腫瘤縮小幅度;劑量依賴性指治療效果會隨劑量提高,而呈現更明顯的改善趨勢,有助於確認有效劑量區間並支持後續劑量選擇。
療效(15 位可評估療效患者)
各劑量組客觀緩解率(ORR)
- 2.56 mg/kg:0%
- 5.12 mg/kg:100%
- 8 mg/kg:81%
- 12 mg/kg:目前觀察到初步 ORR(仍持續收案與追蹤)
疾病控制率(DCR)
- 各劑量組皆為 100%
平均腫瘤縮小幅度
- 2.56 mg/kg:–6.0%
- 5.12 mg/kg:–33.4%
- 8 mg/kg:–46.0%
- 12 mg/kg:–27.9%(數據仍持續成熟)
整體而言,數據顯示療效呈劑量依賴趨勢,腫瘤縮小在中高劑量區間更為顯著;由於 12 mg/kg 組仍在收案及追蹤,其結果目前仍屬初步分析。
安全性
- 在可評估 DLT(劑量限制毒性) 的患者中,未觀察到劑量限制毒性
- 最常見的治療相關不良事件為:
- 白血球數下降 (leukopenia)
- 嗜中性球數下降 (neutropenia)
整體而言,血液學相關不良事件可透過標準支持性治療進行管理。本研究中未觀察到新的安全性訊號。最常見的不良事件(白血球和嗜中性球下降)與化療已知影響一致,整體安全性概況符合預期。
漢康生技集團總裁暨醫療長陸英明博士表示,HCB101 的設計旨在選擇性靶向 CD47–SIRPα 先天免疫檢查點,同時保持與既有腫瘤標準治療方案的高度相容性。在 APGCC (亞太胃食管癌大會) 上,我們報告了 HCB101 在二線胃癌中的明確劑量依賴性活性,包括在 5.12 mg/kg 劑量下確認的腫瘤反應,以及在 8 mg/kg 劑量下更深層的腫瘤縮小。隨著更高劑量組數據持續成熟,這些結果進一步強化我們將 HCB101 發展為差異化先天免疫骨幹療法、並與標準治療無縫整合的策略。
此次APGCC 公布的臨床數據顯示,HCB101 在二線胃癌治療中呈現劑量越高效果越好的趨勢,並支持其與現有癌症標準治療聯合使用的發展方向。
聯合治療機制(Combination Rationale)
此三藥聯合療法旨在於腫瘤微環境中協同多種互補機制:
- HCB101
選擇性阻斷 CD47–SIRPα 先天免疫檢查點,恢復巨噬細胞介導的腫瘤吞噬作用 - Ramucirumab
減少 VEGF 驅動的免疫抑制,並促進腫瘤血管正常化 - Paclitaxel
促進具免疫原性的腫瘤細胞死亡並釋放抗原
三者結合旨在於標準治療架構下,同時增強先天與適應性抗腫瘤免疫反應。
關於 HCB101:新一代 SIRPα Fc 融合蛋白
HCB101 為一種經結構設計開發的 SIRPα–IgG4 Fc 融合蛋白,透過漢康生技的 FBDB™ 平台開發,旨在選擇性阻斷 CD47–SIRPα 先天免疫檢查點並降低血液學毒性。
與早期抗 CD47 療法不同,HCB101 的設計可:
- 保留巨噬細胞介導的抗腫瘤活性
- 降低與紅血球 CD47 的結合
而紅血球結合問題正是過去 CD47 療法發展的一大限制。
透過 AI 輔助結構建模,HCB101 能:
- 對腫瘤細胞 CD47 具有差異化結合能力
- 同時對紅血球 CD47 保持低親和力
其安全性、受體佔據率與藥理特性皆設計為可與既有腫瘤治療方案整合,而不影響標準給藥流程或臨床操作。
在持續進行的臨床與轉譯研究中,HCB101 已展現:
- 持續的靶點結合(target engagement)
- 早期抗腫瘤活性
- 單藥與聯合療法皆具潛力
目前針對胃癌、大腸直腸癌、頭頸癌及三陰性乳癌的劑量爬升與第二期擴展試驗仍在進行中。
關於漢康生技
漢康生技(7827.TPEx)總部位於台北,並於上海與美國舊金山灣區設有據點,是一家全球臨床階段生技公司,專注於腫瘤免疫治療與免疫相關疾病。
公司由具備生物藥發現與全球開發經驗的團隊領導,致力於重新塑造癌症治療的未來。
漢康生技的專有 Fc-based Designer Biologics(FBDB™)平台可設計多功能生物製劑,透過多種靶向模式同時活化先天與適應性免疫途徑,並克服現有 anti-PD1/PD-L1 免疫療法的限制。
FBDB™ 平台已在多個腫瘤動物模型中取得概念驗證數據。漢康生技正推進多項創新生物藥產品管線,以差異化分子設計與可規模化 CMC 製程策略,滿足重大未被滿足的醫療需求。



