HanchorBio Presents First Public Clinical Data for HCB301, a Tri-Specific Innate Checkpoint Molecule, at ESMO-TAT 2026
Initial Phase 1 dose-escalation results demonstrate manageable safety and support continued clinical evaluation of next-generation multi-checkpoint engineering.
[Taipei, Shanghai, San Francisco | March 16, 2026] – HanchorBio, Inc. (TPEx: 7827), a global clinical-stage biotechnology company advancing next-generation immunotherapies for oncology and autoimmune diseases, today announced the presentation of first-in-human clinical data for HCB301 at the ESMO Targeted Anticancer Therapies (TAT) Congress 2026 in Paris, France.
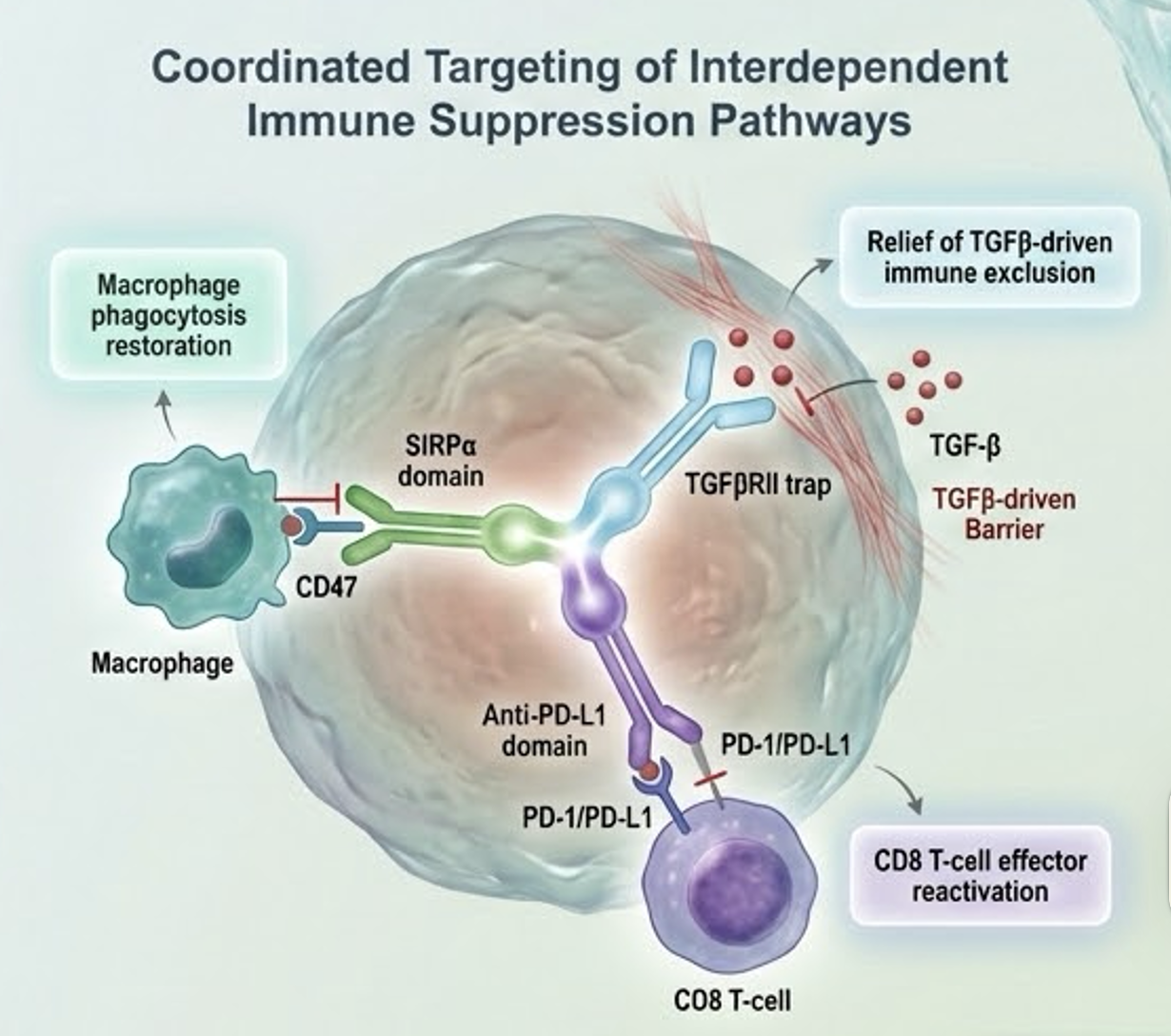
HCB301-101 (NCT06487624) is an ongoing Phase 1 first-in-human study evaluating HCB301, a tri-specific Fc-fusion protein designed to simultaneously target CD47, PD-L1, and TGF-β pathways to coordinate innate, adaptive, and stromal immune modulation. The data were presented in an oral session by the principal investigator, Dr. Ji Zhu, of the Cancer Hospital of the University of Chinese Academy of Sciences (Zhejiang Cancer Hospital).
Presentation Details:
Presentation ID: 600
Title: First-in-Human Phase 1 Evaluation of HCB301, a Tri-specific SIRPα-PD-1-TGFβ Fusion Protein: Safety, Pharmacokinetics, and Multisystem Immune Activation
Session Name / Location: Proffered Paper session / Auditorium Bordeaux, Palais des Congrès
Date / Time: 16 March 2026 / 16:00 – 17:30 CET
Oral Presentation: HCB301-101 Phase 1 Dose Escalation
As of the February 2026 data cutoff:
- 19 patients enrolled across three dose levels (0.3, 0.6, and 1.2 mg/kg)
- Step-up dosing implemented to mitigate infusion-related risk
- Heavily pretreated population (median 4 prior systemic therapies)
- Predominantly Grade 1–2 treatment-related adverse events
- Predictable, on-mechanism cytopenias without a cumulative anemia signal
- No unexpected additive hematologic toxicity despite tri-specific design
Preliminary Clinical Activity:
- 36% disease stabilization (n=14 evaluable) at 0.3–1.2 mg/kg
- Durable stable disease observed across multiple tumor types
- Activity observed below projected full target exposure
These findings support continued dose escalation to further characterize pharmacokinetics, pharmacodynamics, and the exposure-response relationship as the study advances toward defining the recommended Phase 2 dose (RP2D).
“HCB301 is designed to address multi-compartment immune resistance by coordinating SIRPα, PD-L1, and TGF-β modulation within a single molecule,” said Ji Zhu, MD, PhD., Principal Investigator of the HCB301-101 study. “At the early dose levels evaluated to date, we have observed a manageable safety profile with predominantly low-grade cytopenias consistent with the mechanism of action. Importantly, disease stabilization has been observed in a heavily pretreated population with limited therapeutic options, even at exposure levels below projected full target engagement. These early data support continued dose escalation to define the therapeutic window of this tri-specific approach.”
HCB301 is a tri-specific Fc-fusion protein engineered using HanchorBio’s FBDB™ platform to simultaneously modulate SIRPα, PD-L1, and TGF-β pathways within the tumor microenvironment. By coordinating innate immune activation, adaptive checkpoint inhibition, and stromal modulation in a single molecule, HCB301 is designed to address multi-compartment immune resistance while maintaining structural control over hematologic safety parameters.
“HCB301 represents the first clinical validation of our next-generation multi-functional checkpoint architecture,” said Scott Liu, PhD, Founder and Chairman of HanchorBio. “Building on the structural calibration established with HCB101, we selected SIRPα to activate innate phagocytosis, anti-PD-L1 to restore adaptive T-cell function, and TGF-β modulation to address stromal-mediated immune exclusion. The objective was not to increase molecular complexity, but to engineer coordinated modulation across dominant immune-suppression pathways while preserving exposure discipline and hematologic control. These initial clinical findings demonstrate that triple-axis engagement can be advanced with a manageable safety profile, reinforcing the scalability of our FBDB™ platform.”
About HanchorBio
Based in Taipei, Shanghai, and the San Francisco Bay Area, HanchorBio (7827.TPEx) is a global clinical-stage biotechnology company focused on immuno-oncology and immune-mediated diseases. The company is led by an experienced team with a proven track record in biologics discovery and global development, aiming to reshape the landscape of cancer therapies. HanchorBio’s proprietary Fc-based designer biologics (FBDB™) platform enables the design of multi-functional biologics with diverse targeting modalities, designed to activate both innate and adaptive immune pathways and overcome the current challenges of anti-PD1/L1 immunotherapies. The FBDB™ platform has delivered proof-of-concept data in several in vivo tumor animal models. HanchorBio is advancing a portfolio of innovative biologics designed to address significant unmet medical needs through differentiated molecular configurations in R&D and scalable CMC strategies.



