HanchorBio Presents Dose-Dependent Tumor Responses and Deepening Tumor Shrinkage with HCB101 Triple Combination in Second-Line Gastric Cancer at the 11th Asia-Pacific Gastroesophageal Cancer Congress
Early clinical data show 100% ORR at 5.12 mg/kg, 100% DCR across doses, and dose-dependent tumor shrinkage; the 12 mg/kg cohort remains ongoing

HanchorBio, Inc. (TPEx: 7827), a global clinical-stage biotechnology company advancing transformative immunotherapies, today presented new clinical data from its ongoing HCB101-201 Phase 1b/2a study (NCT06771622) at the 11th Asia-Pacific Gastroesophageal Cancer Congress (APGCC) in Okinawa, Japan. The poster highlights early findings supporting dose-dependent antitumor activity of HCB101 in combination with ramucirumab and paclitaxel in patients with advanced gastric adenocarcinoma who progressed after first-line therapy.
The dataset demonstrates dose-dependent tumor reduction and consistent disease control across different dose levels, supporting further evaluation of HCB101 as a differentiated innate immune checkpoint backbone that integrates with standard-of-care regimens.
As previously announced in February 2026, HCB101 has been granted Orphan Drug Designation by the U.S. Food and Drug Administration (FDA) for the treatment of gastric cancer.
Poster Presentation Details
Title: Phase Ib/IIa Study of HCB101 Combination Demonstrates Striking Dose-Dependent Efficacy in Second-Line Gastric Cancer: 100% Partial Responses at 5.12 mg/kg
Presenter: Abigail Yu
Key Findings Highlighted
The poster reports dose-dependent antitumor activity of HCB101 when combined with ramucirumab and paclitaxel in second-line gastric cancer, including both response rates and tumor shrinkage depth.
Efficacy (15 efficacy-evaluable patients across dose levels)
- Objective response rate (ORR) by dose:
-
- 2.56 mg/kg: 0%
- 5.12 mg/kg: 100%
- 8 mg/kg: 81%
- 12 mg/kg (ongoing): preliminary ORR observed to date
- Disease control rate (DCR): 100% across all doses
- Mean tumor shrinkage by dose:
- 2.56 mg/kg: –6.0%
- 5.12 mg/kg: –33.4%
- 8 mg/kg: –46.0%
- 12 mg/kg: –27.9% (data continue to mature)
Overall, the dataset supports a dose-dependent efficacy profile, with tumor reductions deepening through the mid-to-high dose range; interpretation of the 12 mg/kg cohort remains preliminary due to ongoing enrollment and follow-up.
Safety
- No dose-limiting toxicities were observed in DLT-evaluable patients
- The most frequently reported treatment-related adverse events were:
- decreased in white blood cell count (leukopenia)
- decreased in neutrophil count (neutropenia)
Overall, hematologic adverse events were manageable with standard supportive care. No unexpected safety signals were observed in the current study. The most common events (leukopenia and neutropenia) were consistent with the known effects of chemotherapy, and the overall safety profile was consistent with expectations.
“HCB101 is engineered to selectively target the CD47–SIRPα innate immune checkpoint while remaining compatible with established standard oncology regimens,” said Alvin Luk, PhD, MBA, CCRA, President & Chief Medical Officer (Group) and CEO (USA) of HanchorBio. “At APGCC, we reported clear dose-dependent activity in second-line gastric cancer, including confirmed responses at 5.12 mg/kg and deeper tumor shrinkage at 8 mg/kg. As higher-dose cohorts continue to mature, these data reinforce our strategy to develop HCB101 as a differentiated innate immune backbone that integrates seamlessly with standard-of-care therapies.”
Combination Rationale
The regimen is designed to coordinate complementary mechanisms within the tumor microenvironment: HCB101 restores macrophage-mediated phagocytosis by selectively blocking the CD47–SIRPα innate immune checkpoint; ramucirumab mitigates VEGF-driven immunosuppression and supports vascular normalization; and paclitaxel promotes immunogenic tumor cell death and antigen release. Together, the combination aims to enhance both innate and adaptive antitumor responses within a standard-of-care backbone.
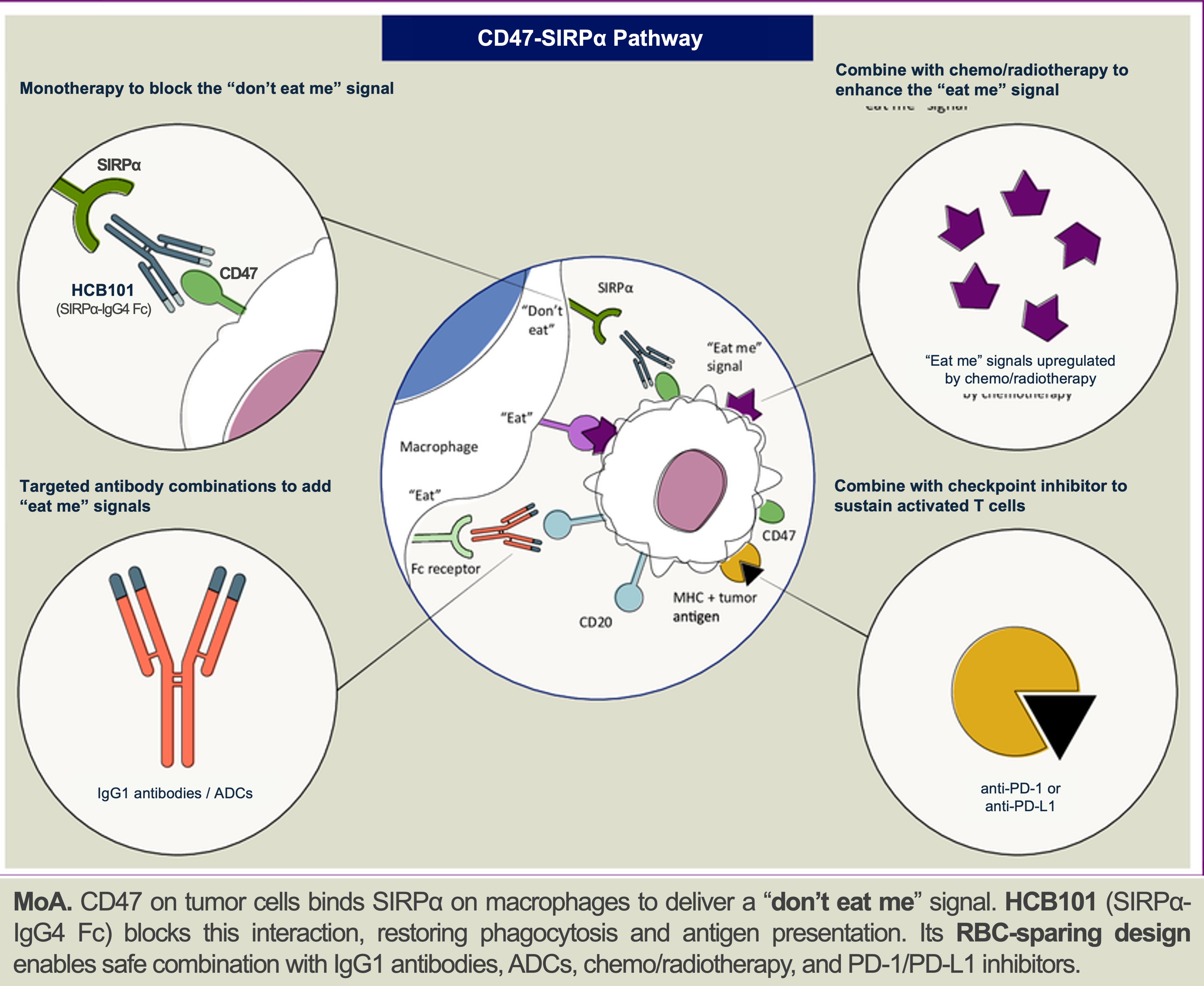
About HCB101: A Next-Generation SIRPα Fc-Fusion Protein
HCB101 is a rationally engineered SIRPα–IgG4 Fc fusion protein developed using HanchorBio’s FBDB™ platform to selectively block the CD47–SIRPα innate immune checkpoint while minimizing hematologic toxicity. Unlike earlier anti-CD47 approaches, HCB101 is designed to preserve macrophage-mediated antitumor activity while reducing red blood cell binding, a limitation that historically constrained CD47-directed therapies.
Engineered using AI-assisted structural modeling, HCB101 achieves differentiated binding to CD47 on tumor cells while maintaining low affinity for CD47 on red blood cells. Its safety, receptor occupancy, and pharmacologic characteristics are designed to enable integration with established oncology regimens without disrupting standard dosing or clinical workflows.
In ongoing clinical and translational evaluation, HCB101 has demonstrated sustained target engagement and early antitumor activity as both monotherapy and in combination, including in tumor types historically challenging for CD47-directed therapies. Dose escalation and Phase 2 expansion cohorts in gastric, colorectal, head-and-neck, and triple-negative breast cancers are ongoing.
These attributes support HCB101’s development as a differentiated innate immune checkpoint backbone with broad potential across solid tumors and hematologic malignancies.
About HanchorBio
Based in Taipei, Shanghai, and the San Francisco Bay Area, HanchorBio (7827.TPEx) is a global clinical-stage biotechnology company focused on immuno-oncology and immune-mediated diseases. The company is led by an experienced team with a proven track record in biologics discovery and global development, aiming to reshape the landscape of cancer therapies. HanchorBio’s proprietary Fc-based designer biologics (FBDB™) platform enables the design of multi-functional biologics with diverse targeting modalities, designed to activate both innate and adaptive immune pathways and overcome the current challenges of anti-PD1/L1 immunotherapies. The FBDB™ platform has delivered proof-of-concept data in several in vivo tumor animal models. HanchorBio is advancing a portfolio of innovative biologics designed to address significant unmet medical needs through differentiated molecular configurations in R&D and scalable CMC strategies.



