HanchorBio Presents First Public Clinical Data for HCB301, a Tri-Specific Innate Checkpoint Molecule, at ESMO-TAT 2026
Initial Phase 1 dose-escalation results demonstrate manageable safety and support continued clinical evaluation of next-generation multi-checkpoint engineering.
[Taipei, Shanghai, San Francisco | March 16, 2026] – HanchorBio, Inc. (TPEx: 7827), a global clinical-stage biotechnology company advancing next-generation immunotherapies for oncology and autoimmune diseases, today announced the presentation of first-in-human clinical data for HCB301 at the ESMO Targeted Anticancer Therapies (TAT) Congress 2026 in Paris, France.
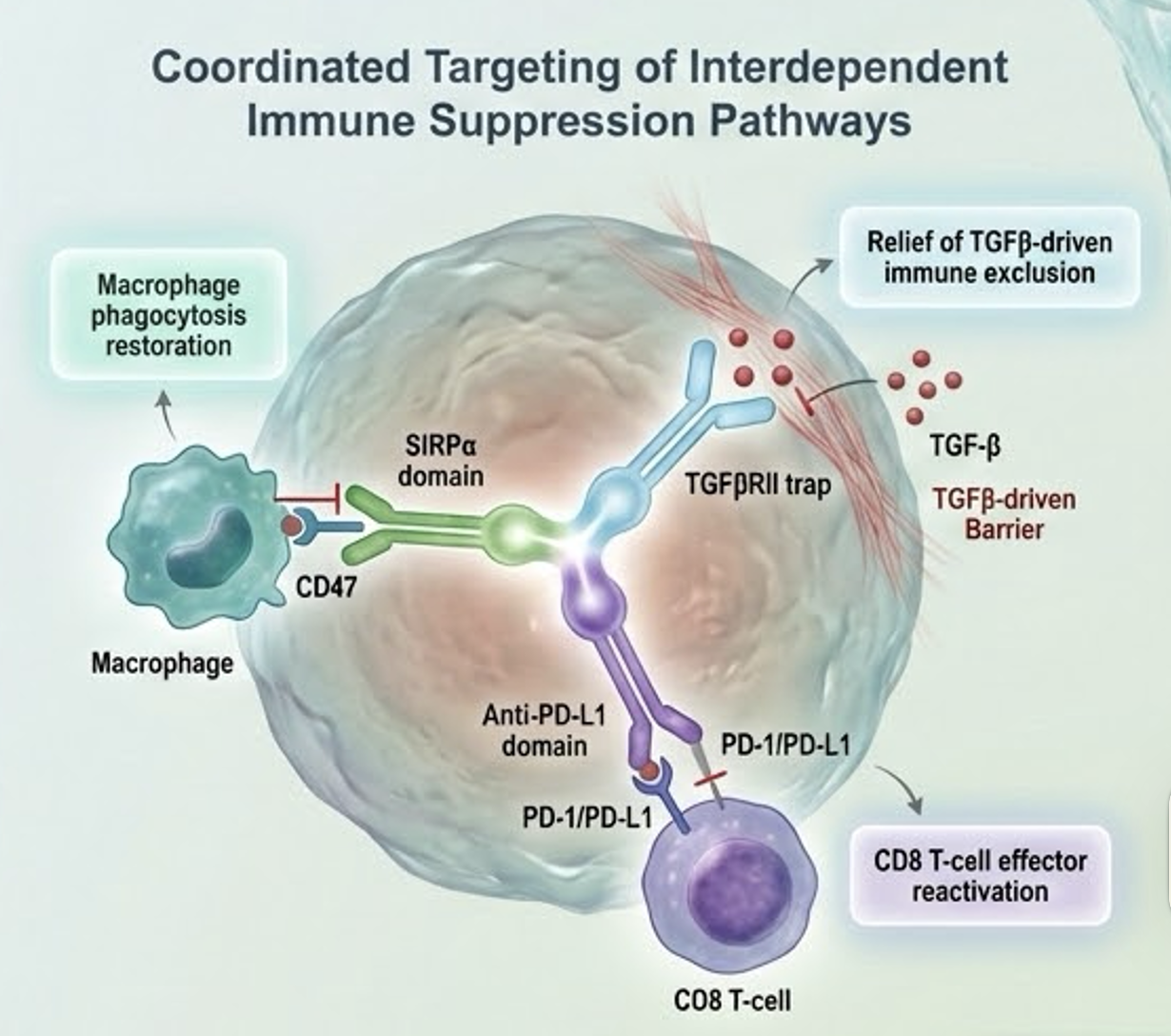
HCB301-101 (NCT06487624) is an ongoing Phase 1 first-in-human study evaluating HCB301, a tri-specific Fc-fusion protein designed to simultaneously target CD47, PD-L1, and TGF-β pathways to coordinate innate, adaptive, and stromal immune modulation. The data were presented in an oral session by the principal investigator, Dr. Ji Zhu, of the Cancer Hospital of the University of Chinese Academy of Sciences (Zhejiang Cancer Hospital).
Presentation Details:
Presentation ID: 600
Title: First-in-Human Phase 1 Evaluation of HCB301, a Tri-specific SIRPα-PD-1-TGFβ Fusion Protein: Safety, Pharmacokinetics, and Multisystem Immune Activation
Session Name / Location: Proffered Paper session / Auditorium Bordeaux, Palais des Congrès
Date / Time: 16 March 2026 / 16:00 – 17:30 CET
Oral Presentation: HCB301-101 Phase 1 Dose Escalation
As of the February 2026 data cutoff:
- 19 patients enrolled across three dose levels (0.3, 0.6, and 1.2 mg/kg)
- Step-up dosing implemented to mitigate infusion-related risk
- Heavily pretreated population (median 4 prior systemic therapies)
- Predominantly Grade 1–2 treatment-related adverse events
- Predictable, on-mechanism cytopenias without a cumulative anemia signal
- No unexpected additive hematologic toxicity despite tri-specific design
Preliminary Clinical Activity:
- 36% disease stabilization (n=14 evaluable) at 0.3–1.2 mg/kg
- Durable stable disease observed across multiple tumor types
- Activity observed below projected full target exposure
These findings support continued dose escalation to further characterize pharmacokinetics, pharmacodynamics, and the exposure-response relationship as the study advances toward defining the recommended Phase 2 dose (RP2D).
“HCB301 is designed to address multi-compartment immune resistance by coordinating SIRPα, PD-L1, and TGF-β modulation within a single molecule,” said Ji Zhu, MD, PhD., Principal Investigator of the HCB301-101 study. “At the early dose levels evaluated to date, we have observed a manageable safety profile with predominantly low-grade cytopenias consistent with the mechanism of action. Importantly, disease stabilization has been observed in a heavily pretreated population with limited therapeutic options, even at exposure levels below projected full target engagement. These early data support continued dose escalation to define the therapeutic window of this tri-specific approach.”
HCB301 is a tri-specific Fc-fusion protein engineered using HanchorBio’s FBDB™ platform to simultaneously modulate SIRPα, PD-L1, and TGF-β pathways within the tumor microenvironment. By coordinating innate immune activation, adaptive checkpoint inhibition, and stromal modulation in a single molecule, HCB301 is designed to address multi-compartment immune resistance while maintaining structural control over hematologic safety parameters.
“HCB301 represents the first clinical validation of our next-generation multi-functional checkpoint architecture,” said Scott Liu, PhD, Founder and Chairman of HanchorBio. “Building on the structural calibration established with HCB101, we selected SIRPα to activate innate phagocytosis, anti-PD-L1 to restore adaptive T-cell function, and TGF-β modulation to address stromal-mediated immune exclusion. The objective was not to increase molecular complexity, but to engineer coordinated modulation across dominant immune-suppression pathways while preserving exposure discipline and hematologic control. These initial clinical findings demonstrate that triple-axis engagement can be advanced with a manageable safety profile, reinforcing the scalability of our FBDB™ platform.”
About HanchorBio
Based in Taipei, Shanghai, and the San Francisco Bay Area, HanchorBio (7827.TPEx) is a global clinical-stage biotechnology company focused on immuno-oncology and immune-mediated diseases. The company is led by an experienced team with a proven track record in biologics discovery and global development, aiming to reshape the landscape of cancer therapies. HanchorBio’s proprietary Fc-based designer biologics (FBDB™) platform enables the design of multi-functional biologics with diverse targeting modalities, designed to activate both innate and adaptive immune pathways and overcome the current challenges of anti-PD1/L1 immunotherapies. The FBDB™ platform has delivered proof-of-concept data in several in vivo tumor animal models. HanchorBio is advancing a portfolio of innovative biologics designed to address significant unmet medical needs through differentiated molecular configurations in R&D and scalable CMC strategies.
漢康生技於 ESMO標靶抗癌治療大會首次公開 HCB301 人體臨床數據,展現三功能先天免疫檢查點分子的臨床開發潛力
一期臨床試驗初步劑量遞增數據顯示具可控的安全性,支持新一代多靶點/多功能的工程設計持續推進臨床開發
漢康生技為全球臨床階段生技公司,專注於開發腫瘤與自體免疫疾病的次世代免疫療法,在 2026 年法國巴黎舉行的 ESMO標靶抗癌治療大會(ESMO Targeted Anticancer Therapies Congress)首次公開發表 HCB301 的人體臨床數據。
HCB301-101(NCT06487624)為一項持續進行中的一期人體臨床試驗,評估 HCB301 的安全性與初步臨床特性。HCB301 為一種三功能、三靶點基於 Fc設己的融合蛋白創新生物藥,設計上可同時作用於 CD47、PD-L1 與 TGF-β 路徑,以協同調節先天免疫、適應性免疫與腫瘤微環境。本次數據由中國醫學科學院腫瘤醫院(浙江省腫瘤醫院)研究主持人朱驥博士於口頭報告中發表。
發表資訊
- 發表編號: 600
- 題目: First-in-Human Phase 1 Evaluation of HCB301, a Tri-specific SIRPα-PD-1-TGFβ Fusion Protein: Safety, Pharmacokinetics, and Multisystem Immune Activation
- 場次/地點: 法國巴黎Palais des Congrès
- 日期/時間: 2026 年 3 月 16 日 16:00–17:30 CET
口頭發表:HCB301-101 第一劑量遞增研究
截至 2026 年 2 月資料截止日,HCB301-101 研究共納入 19 位患者,涵蓋三個劑量層級(0.3、0.6 與 1.2 mg/kg)。研究採用 step-up dosing 給藥策略,以降低輸注相關風險。受試者皆為經多線治療之患者,先前系統性治療中位數為 4 線。
整體安全性結果顯示,治療相關不良事件以第 1 至 2 級為主,並觀察到可預期、與作用機制一致的血球減少現象,但未見累積性貧血訊號;儘管 HCB301 為三功能設計,亦未觀察到非預期的加成性血液學毒性。
初步臨床活性
在 14 位可評估患者中,於 0.3–1.2 mg/kg 劑量區間觀察到 36% 的疾病穩定率。此外,多種腫瘤類型中皆觀察到持久性疾病穩定,且在尚未達到預估完整標的暴露的情況下,即已觀察到初步活性訊號。
上述結果支持研究持續進行劑量遞增,以進一步釐清藥物動力學、藥效學以及暴露量與反應之間的關聯,並朝建議第二期試驗劑量(RP2D)推進。
HCB301-101 研究主持人朱驥醫學博士表示:「HCB301 的設計目標,是在單一分子中整合 SIRPα、PD-L1 與 TGF-β 的調節作用,以對抗腫瘤免疫抗性涉及的多重區室機制。就目前已評估的早期劑量而言,我們觀察到HCB301整體安全性可控,觀察到的血球減少多為低等級,且與其作用機制相符。更重要的是,即使在尚未達到預期完整標的結合的暴露條件下,仍在多線治療後、治療選擇有限的患者族群中觀察到疾病穩定。這些初步結果支持研究持續進行劑量遞增,以進一步確認此三特異性療法的適當治療窗。」
關於 HCB301:三特異性免疫檢查點療法
HCB301 是漢康生技的新一代免疫療法,旨在將三種協同機制整合於單一分子中:阻斷 CD47-SIRPα 以激活髓系細胞的吞噬作用、抑制 PD-1 以恢復耗竭的 T 細胞功能,以及抑制 TGF-β 通路以對抗免疫逃逸。HCB301 同樣採用專有的 FBDB™ 平台開發,代表了多重檢查點免疫療法的新一代研發趨勢。臨床前研究已證實其具有增強免疫激活與強效抗腫瘤的能力,相關研究結果先前已於 2025 年癌症免疫治療學會(SITC)年會發表。
漢康生技創辦人暨董事長劉世高博士表示:「HCB301 是我們新一代多功能檢查點架構首次進入臨床驗證的重要成果。延續 HCB101 所建立的結構校準基礎,我們選擇以 SIRPα 活化先天免疫吞噬作用、以 anti-PD-L1 恢復適應性 T 細胞功能,並透過 TGF-β 調節來改善腫瘤基質介導的免疫排斥。我們的目標並非單純提高分子複雜度,而是希望在維持暴露控制與血液學安全性的前提下,對主要免疫抑制路徑進行協同調節。這次初步臨床結果顯示,三軸機制的整合推進具備可控的安全性,也再次驗證 FBDB™ 平台的延展性。」
關於漢康生技
漢康生技(證券代碼:7827.TPEx)總部位於台北、上海與舊金山灣區,是一家全球臨床階段生技公司,專注於腫瘤免疫治療與免疫相關疾病新藥開發。公司由具備生物藥研發與全球開發實績的團隊領導,致力於透過創新分子設計重塑癌症治療格局。
漢康生技所開發的 Fc-based designer biologics(FBDB™)平台可設計多功能生物藥,透過多元標的組合,同步激活先天與後天免疫路徑,並克服現有 anti-PD1/L1 免疫療法所面臨的挑戰。透過在多功能生物藥研發領域的突破,以及具備高擴展性的 CMC(化學製造與管制)策略,漢康生技正加速推進創新藥物管線,以解決尚未被滿足的重大醫療需求。



